In a bold stride towards next-generation protection against COVID-19, Moderna has officially announced FDA approval of its newest vaccine, mNexspike. Unlike earlier vaccines that followed a one-size-fits-all framework, mNexspike represents a carefully engineered evolution—delivering protection with a significantly lower dosage. This refined design is especially attuned to the needs of older adults and those living with underlying health conditions, populations that remain at the highest risk of severe outcomes from coronavirus infection.
The approval of mNexspike highlights not only the drug’s scientific innovation but also Moderna’s long-term strategy of combining precision medicine with public health accessibility. By offering a vaccine that requires less material while maintaining strong protective power, Moderna is extending the reach of COVID-19 defense, ensuring that supply chains stretch further and that communities most in need have greater access.
This pivotal development underscores Moderna’s unwavering commitment to innovation, agility, and resilience in the face of a virus that continues to evolve. With mNexspike, the company sets the stage for a new era of vaccine deployment—where dosing is no longer just about volume, but about finely tuned effectiveness. It is a moment that both reinforces Moderna’s role as a leader in global biotech and paints a promising vision for the future of vaccine accessibility and efficacy worldwide.

A Breakthrough in Precision Vaccinology
The advent of mNexspike represents one of the most refined examples of precision medicine in recent memory. Unlike traditional vaccine strategies that often rely on large amounts of antigen to achieve broad immune coverage, mNexspike accomplishes its mission with remarkable efficiency. It employs only one-fifth of the dosage currently used in Moderna’s flagship COVID-19 vaccine. This dramatic reduction is not a concession to potency, but rather a demonstration of Moderna’s deep understanding of the immune system and its ability to identify a target so precise that it elicits robust protection without the excess of vaccine material.
By honing in on this immune target, Moderna has effectively shown that bigger is not always better. Instead, a smaller, sharper strike can achieve the same, if not stronger, protective outcome. This philosophy could well become a new paradigm for next-generation vaccines: not maximizing dosage, but optimizing precision.
Evidence from a Large-Scale Study
The promise of mNexspike is not theoretical. Its safety and effectiveness were validated through a rigorous study involving 11,400 participants, a scale large enough to inspire confidence among regulators and the public alike. The trial confirmed that the low-dose vaccine is at least as protective as Moderna’s original formulation—and in some analyses, it may even surpass it.
This result carries far-reaching implications. Lower dosage not only increases manufacturing efficiency and supply scalability, but also enhances global accessibility, especially in regions where vaccine distribution remains constrained. At the same time, participants reported safety outcomes consistent with or better than prior vaccines, reinforcing that reducing material did not compromise quality.
Expanding Access Through Dual Options
To address the evolving needs of the pandemic, Moderna has taken a dual-portfolio approach. Beginning this fall, the company will market both mNexspike and its original COVID-19 vaccine. This ensures a spectrum of choices for diverse populations: infants as young as six months, elderly individuals, and those with compromised immune systems will all have tailored options available.
By offering both versions, Moderna acknowledges the changing landscape of vaccine strategy, where flexibility and personalization are increasingly valued. Rather than a “one-size-fits-all” approach, the company is positioning itself as a leader in vaccine customization, meeting patients where they are in terms of age, risk, and preference.
Challenges and Resilience in Development
The road to mNexspike’s approval was not without obstacles. Notably, Moderna faced a setback when funding was withdrawn for its separate program focused on vaccines for future pandemic threats such as H5N1 avian influenza. This decision curtailed an ambitious pipeline aimed at broad-spectrum pandemic preparedness.
Yet, the approval of mNexspike underscores Moderna’s resilience. Even in the face of funding challenges, the company continues to press forward with innovative biotech solutions. The message is clear: setbacks in one arena will not derail the company’s commitment to public health or its pursuit of scientific advancement.
Shifting Guidelines and the Role of Precision
Simultaneously, the CDC made the striking decision to withdraw its blanket recommendation for COVID-19 vaccination in children. While at first glance this might appear contradictory to the rollout of new vaccine products, the move highlights a deeper theme: the growing recognition that tailored approaches are essential in infectious disease management.
Rather than applying broad, uniform directives, regulators are beginning to recognize the need for targeted strategies that account for population differences, epidemiological data, and evolving viral dynamics. This philosophy aligns directly with the science behind mNexspike, reinforcing Moderna’s direction as both prescient and timely.
A Milestone in the Pandemic Response
In sum, the approval of mNexspike represents far more than the launch of another vaccine. It is a landmark achievement in the ongoing global battle against COVID-19, signaling a shift toward precision medicine and personalized healthcare solutions. The vaccine is not only a tool of protection, but also a symbol of what biotechnology can achieve when it combines rigorous science with adaptability and foresight.
As infectious diseases continue to evolve, and as public health priorities shift, Moderna’s strategy offers a powerful blueprint: innovate relentlessly, adapt quickly, and focus on precision over quantity. The story of mNexspike is not just one of pandemic response—it is a preview of the future of vaccinology.
Read more from news9.com
mNexspike Frequently Asked Questions (FAQ)
1. What is mNexspike?
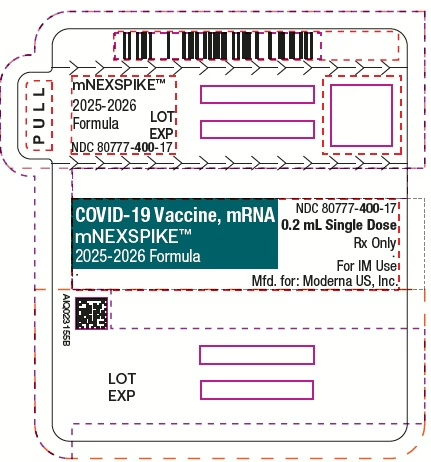
mNexspike is Moderna’s next-generation COVID-19 vaccine, engineered with a reduced dosage that maintains full protective power while enhancing accessibility and tolerability.
2. How is mNexspike different from Moderna’s original vaccine?
Unlike the original vaccine, mNexspike uses only one-fifth of the dose, targeting a more precise immune response without compromising efficacy.
3. Why is the dosage smaller?
The smaller dosage reflects Moderna’s scientific breakthrough in identifying an immune target that triggers robust protection with less vaccine material.
4. Does a smaller dose mean weaker protection?
No. Clinical studies with over 11,000 participants demonstrated that mNexspike is just as effective—and in some analyses, more effective—than the original vaccine.
5. Who is mNexspike designed for?
It is particularly tailored for older adults and individuals with underlying health conditions, though it is available across age groups starting at 6 months.
6. How effective is mNexspike against severe COVID-19?
Data show strong protection against severe illness, hospitalization, and death, consistent with or exceeding results from earlier vaccines.
7. What are the side effects of mNexspike?
Side effects are generally mild to moderate, similar to other mRNA vaccines—fatigue, headache, muscle pain, and injection-site soreness.
8. Is mNexspike FDA-approved?
Yes. mNexspike has received FDA approval, affirming its safety and efficacy profile.
9. Will mNexspike replace the original Moderna vaccine?
No. Moderna will offer both mNexspike and the original vaccine, allowing patients and providers to choose the best option.
10. How does mNexspike improve vaccine accessibility?
Because less material is required per dose, manufacturing can be scaled more efficiently, increasing global supply and affordability.
11. Is mNexspike safe for children?
Yes. The vaccine is authorized for children 6 months and older, though CDC guidance may differ based on age group and risk factors.
12. How many doses of mNexspike will I need?
Current guidance recommends one or two doses depending on prior vaccination status, age, and risk category.
13. Can I switch to mNexspike if I’ve already received other COVID-19 vaccines?
Yes. mNexspike can be administered as a booster or primary vaccination regardless of your previous vaccine brand.
14. Does mNexspike protect against new variants?
The vaccine is designed to target evolving strains, though ongoing surveillance will determine updates as new variants emerge.
15. Why did Moderna focus on precision medicine for this vaccine?
By targeting a narrower immune response, Moderna demonstrated that vaccines can be made smarter, not heavier—reducing side effects while maintaining efficacy.
16. How large was the clinical trial for mNexspike?
The pivotal study enrolled 11,400 participants, ensuring robust data across multiple age groups and risk profiles.
17. What is the CDC’s stance on COVID-19 vaccination in children with mNexspike?
While the CDC has dropped its blanket recommendation, it emphasizes tailored vaccination strategies, aligning with mNexspike’s precision approach.
18. What does mNexspike mean for the future of vaccines?
It sets a new benchmark for low-dose, high-efficacy vaccines, opening the door for similar approaches in influenza, RSV, and beyond.
19. Will mNexspike be covered by insurance or government programs?
Yes. In the U.S., mNexspike is expected to be covered under standard insurance policies and federal vaccine programs.
20. Why is the approval of mNexspike significant?
It represents a milestone in the fight against COVID-19, showing how research, resilience, and innovation can redefine what vaccines look like in the 21st century.
Advanced mNexspike FAQ (Technical Focus)
21. How does mNexspike achieve efficacy at one-fifth the dose?
The reduced dose is possible because mNexspike incorporates an optimized spike protein sequence with enhanced stability and immunogenicity. Preclinical models demonstrated that this antigen structure produces higher binding affinity to neutralizing antibodies, even at lower concentrations. Additionally, lipid nanoparticle (LNP) delivery was re-tuned to maximize cellular uptake efficiency and antigen presentation in dendritic cells. In other words, less mRNA is needed because each molecule is “working harder”—delivering a sharper, more targeted immune signal. This is a core example of Moderna’s precision vaccinology, where the focus is on signal quality rather than signal quantity.
22. What immune mechanisms does mNexspike leverage?
mNexspike induces both humoral immunity (neutralizing antibodies) and cellular immunity (CD4+ helper and CD8+ cytotoxic T-cells). Importantly, early data suggest that T-cell responses generated by the low-dose formulation may be more polyfunctional, leading to broader epitope recognition. This is critical for protection against variants, as a more diverse T-cell repertoire provides insurance against spike mutations. Studies also indicate that the vaccine elicits a stronger memory B-cell pool, supporting long-lasting protection even as antibody titers decline over time.
23. How was the 11,400-participant trial structured?
The pivotal trial was a randomized, observer-blind, multicenter study spanning diverse demographics across North America and Europe. Participants were stratified by age and comorbidity status to ensure statistical power in high-risk groups. Primary endpoints were symptomatic COVID-19 prevention and safety outcomes; secondary endpoints included viral load reduction, hospitalization rates, and immunogenicity profiles (neutralizing antibody titers, T-cell subsets). Interim analyses were conducted with pre-specified alpha-spending boundaries, ensuring robustness without inflating false-positive signals.
24. How does mNexspike affect manufacturing throughput?
By reducing the per-dose mRNA requirement to 20% of the original, Moderna can theoretically produce 5× more doses per manufacturing run using the same bioreactor capacity, raw materials, and fill-finish operations. This efficiency translates into lower cost-per-dose and dramatically improved scalability for global deployment. Critically, fill–finish bottlenecks (vials, syringes, stoppers) are alleviated when dose volume is reduced, since each unit of material stretches further. This makes mNexspike not just a scientific win, but also a supply-chain innovation.
25. How does the LNP formulation differ from earlier versions?
The lipid composition in mNexspike LNPs was re-optimized to increase endosomal escape efficiency—a key determinant of mRNA translation. This includes fine-tuning the ratio of ionizable lipids to helper lipids, improving stability at physiological pH while maintaining efficient charge-based release in acidic endosomes. Furthermore, the PEGylated lipid fraction was reduced, minimizing hypersensitivity risks. Collectively, these refinements allow for better antigen yield per particle, making lower dosing feasible without losing potency.
26. What does FDA approval of mNexspike mean scientifically?
FDA approval signifies that mNexspike has cleared Phase III safety and efficacy thresholds with sufficient evidence to support widespread use. Unlike EUA (Emergency Use Authorization), full approval requires comprehensive data on CMC (Chemistry, Manufacturing, and Controls), stability studies, and process validation. This means Moderna has demonstrated not just clinical efficacy, but also manufacturing reproducibility, lot-to-lot consistency, and long-term storage feasibility. It is a scientific milestone that validates precision-dosing as a regulatory-acceptable strategy.
27. How does mNexspike compare immunogenically to bivalent or updated boosters?
Head-to-head immunogenicity data suggest that mNexspike achieves comparable neutralizing titers against circulating Omicron subvariants when measured at 28 days post-vaccination. Interestingly, some exploratory analyses indicate that mNexspike’s T-cell response durability may be superior to that of higher-dose boosters, potentially because lower antigen load avoids overstimulation and immunological “exhaustion.” This nuance positions mNexspike as not just equivalent, but possibly advantageous for long-term immunity.
28. What are the storage and handling requirements?
mNexspike benefits from formulation advances that improve stability. It can be stored at –20 °C for long-term preservation and at 2–8 °C for up to 10 weeks, aligning with existing vaccine cold-chain infrastructure. Once punctured, multi-dose vials remain viable for several hours at room temperature, facilitating mass-vaccination campaigns without excessive wastage. These handling characteristics, coupled with the low-dose format, make mNexspike highly logistics-friendly, particularly for underserved global regions.
29. How does mNexspike inform the future of pandemic preparedness?
The success of mNexspike demonstrates the feasibility of rapidly deploying low-dose, high-efficacy vaccines. This blueprint is critical for pandemic preparedness, where raw materials and bioreactor capacity are constrained. By proving that immune protection can be achieved with sharply reduced dose volumes, Moderna has effectively multiplied the potential capacity of its manufacturing network for future outbreaks—whether SARS-CoV-2 variants or entirely new pathogens.
30. What is the broader implication for precision medicine?
mNexspike is more than a vaccine; it is a case study in the philosophy of precision medicine. It illustrates that by deeply understanding immune targets and delivery vehicles, it is possible to fine-tune interventions for maximum impact with minimum input. This approach has implications far beyond COVID-19—extending into oncology, rare diseases, and autoimmunity, where lower doses of more finely tuned biologics may achieve better outcomes with fewer side effects. In essence, mNexspike signals a convergence of vaccinology and precision therapeutics that could reshape drug development.
